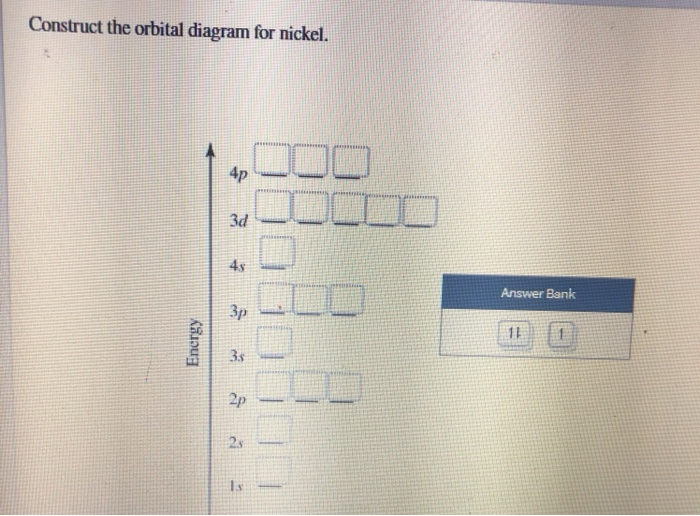
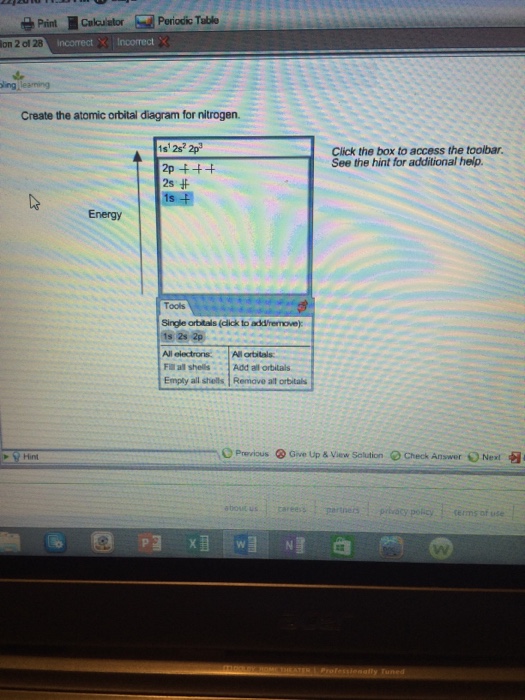
37 create the atomic orbital diagram for nitrogen.
This photo about: Atomic orbital Diagram Nitrogen, entitled as Create The Atomic Orbital Diagram For Nitrogen – Molecules Free Atomic Orbital Diagram Nitrogen - also describes Create The Atomic Orbital Diagram For Nitrogen – Molecules Free and labeled as: atomic orbital band structure,atomic orbital ground state,atomic orbital hybridization pdf,atomic orbital nitrogen,atomic orbital p ... Figure 8.2. 11: The four valence atomic orbitals from an isolated carbon atom all hybridize when the carbon bonds in a molecule like CH 4 with four regions of electron density. This creates four equivalent sp 3 hybridized orbitals. Overlap of each of the hybrid orbitals with a hydrogen orbital creates a C-H σ bond.
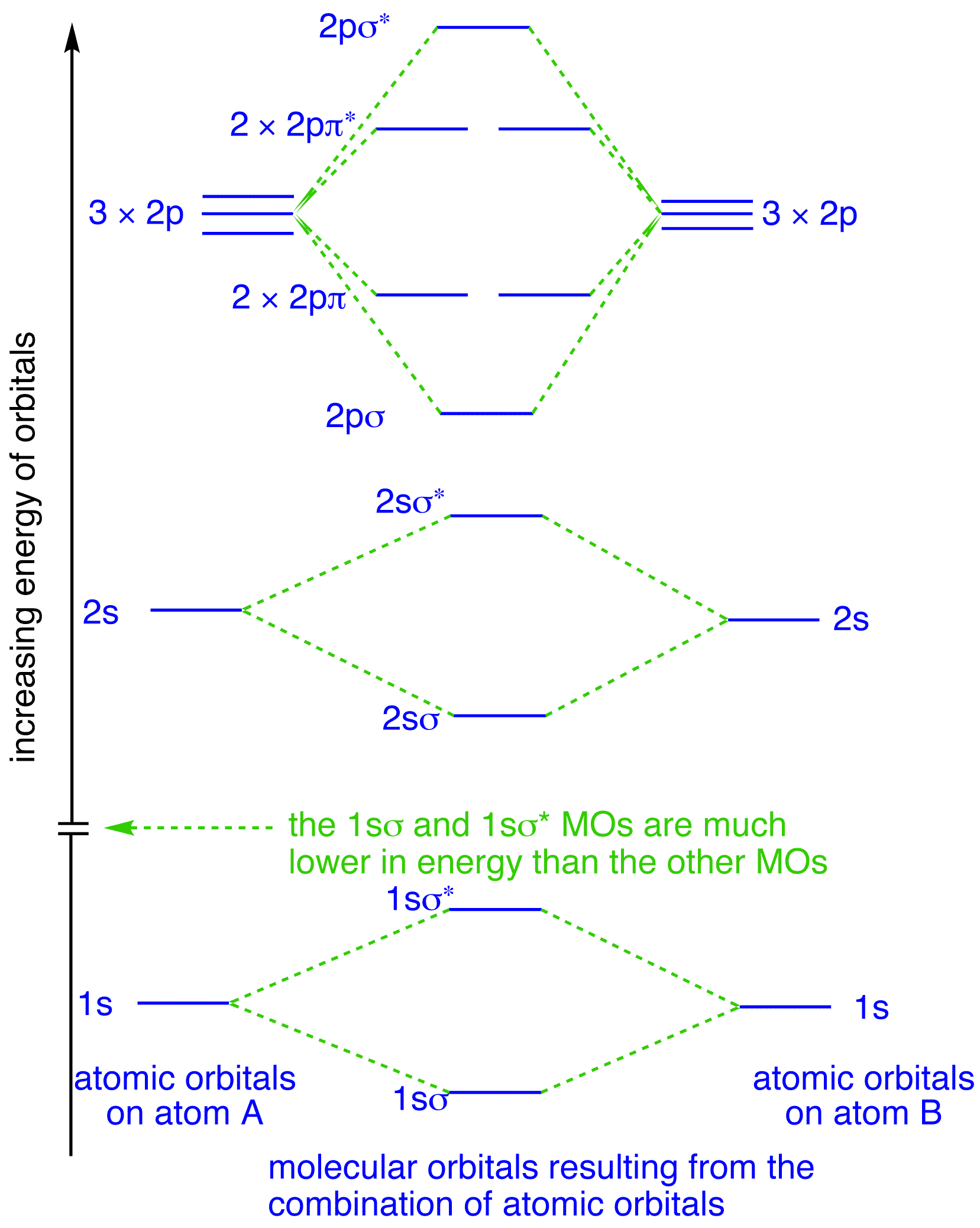
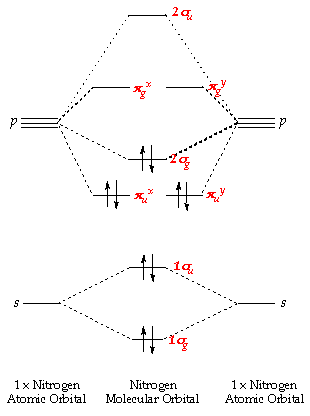
Molecular orbitals in Nitrogen. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. There are four molecular orbitals derived from the 1s and 2s orbitals. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The p orbitals combine to produce a sigma and two ...

Create the atomic orbital diagram for nitrogen.
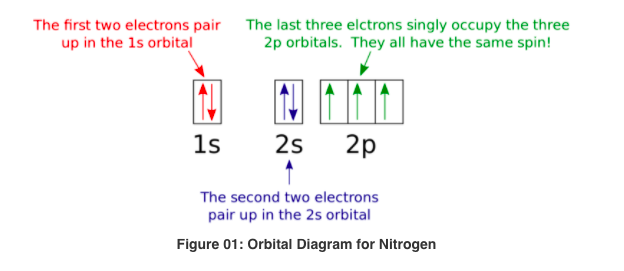
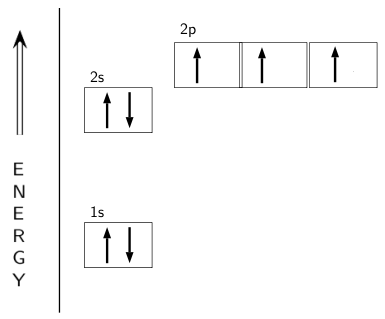
The choice A accurately specifies and illustrates the orbital diagram of a Nitrogen atom with 7 electrons. Based on the number of electrons in a Nitrogen atom, there are two energy levels, the s and p sub-levels: Nitrogen = 2, 5 . The first energy level, S will take up two electrons with opposite spin. What is the atomic orbital diagram for nitrogen? The remaining three electrons will go in the 2p orbital. Therefore the N electron configuration will be 1s22s22p3. The configuration notation for Nitrogen (N) provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of the Nitrogen atom. Create the atomic orbital Diagram for Nitrogen. solved create the atomic orbital diagram for nitrogen st answer to create the atomic orbital diagram for nitrogen start by adding the appropriate subshells for example boron is in the create the atomic orbital diagram for nitrogen in some cases we may need to slightly alter the design color or even accessories we want a new thought for it then ...
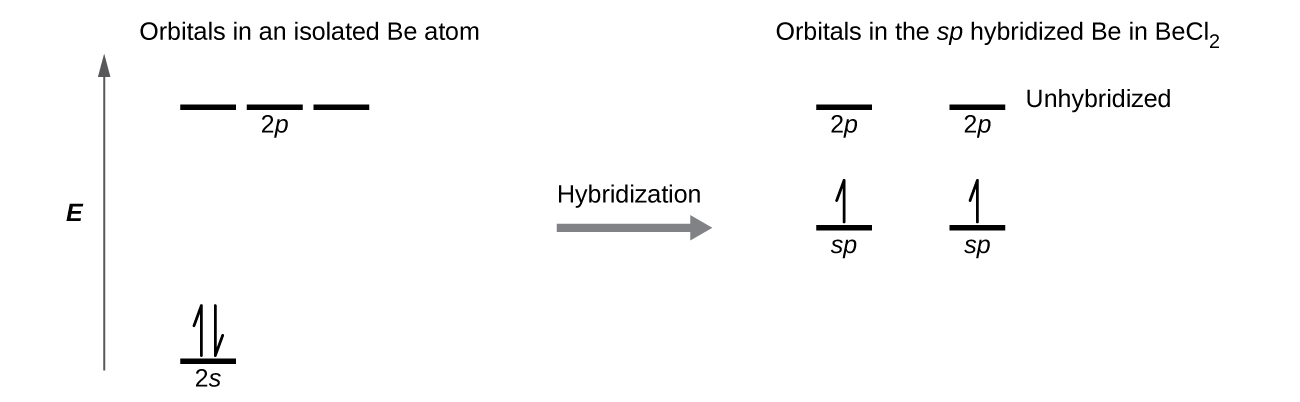
Create the atomic orbital diagram for nitrogen.. Create the atomic orbital diagram for nitrogen. Learn this topic by watching The Electron Configuration Concept Videos All Chemistry Practice Problems The Electron Configuration Practice Problems In writing the electron configuration for nitrogen the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for N goes in the 2s orbital. The remaining three electrons will go in the 2p orbital. Therefore the N electron configuration will be 1s 2 2s 2 2p 3. If playback doesn't begin shortly ... This orbital energy-level diagram shows the sp hybridized orbitals on Be in the linear BeCl 2 molecule. Each of the two sp hybrid orbitals holds one electron and is thus half filled and available for bonding via overlap with a Cl 3p orbital. When atomic orbitals hybridize, the valence electrons occupy the newly created orbitals. If that single electron were a spin-up (ms = +1/2), the orbital diagram for The figure below illustrating orbital diagrams for nitrogen is similar to the. Use orbital filling diagrams to describe the locations of electrons in an atom. Diagram of Hund's rule in boron, carbon, nitrogen, and oxygen. Figure 1. The 2p .
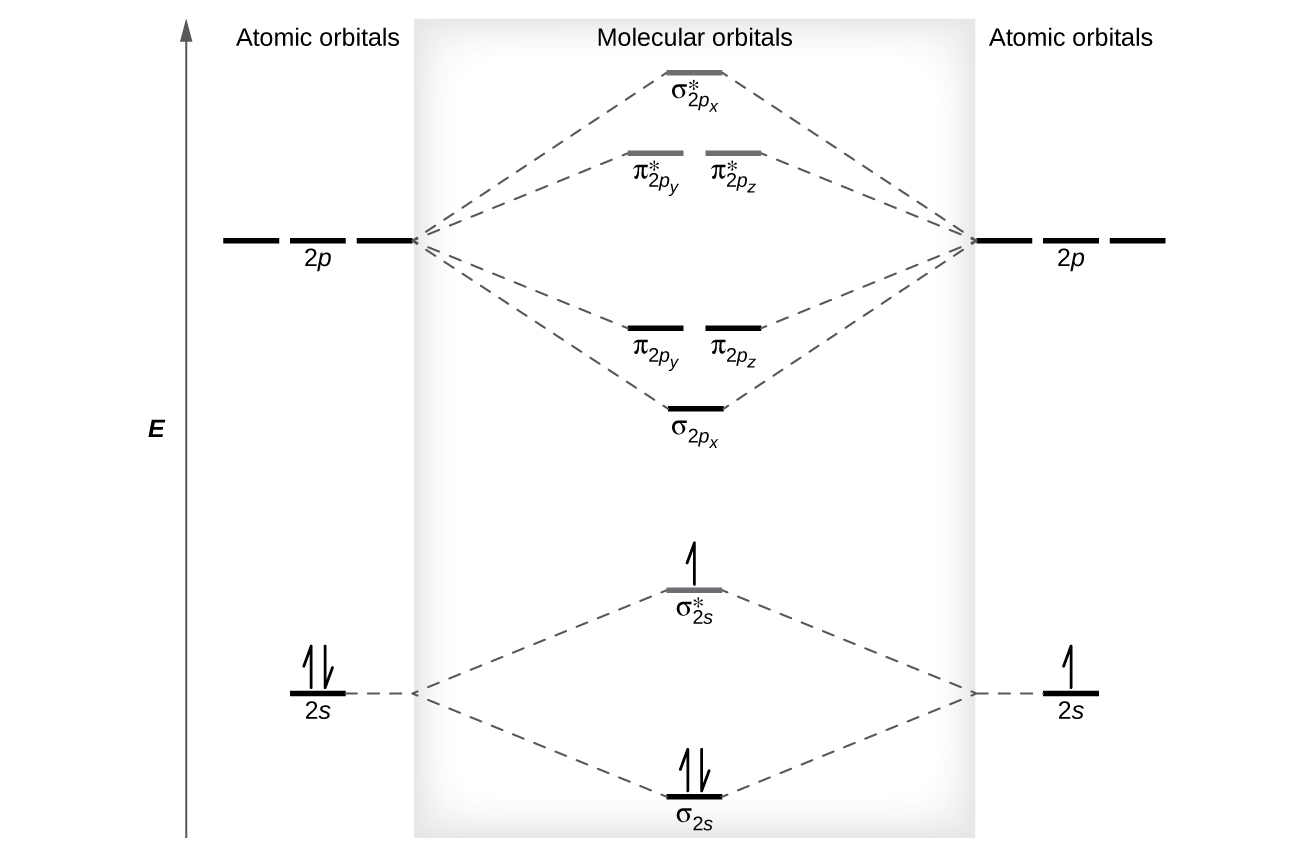
Atomic orbital Diagram Nitrogen. molecular orbital diagram a molecular orbital diagram or mo diagram is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory molecular orbital theory home faculty molecular orbital theory the goal of molecular orbital theory is to describe molecules in a similar way to how we describe atoms that is in Which nitrogen atom(s) is/are sp 3 hybridized. 5. Describe the bonding scheme of CH 4. Answers: 1. a and b. 2. Just like the energy diagram in fig.3. For carbon, each sp 3 orbital has 1 electron. For nitrogen, the first sp 3 orbital has 2 electrons, then one electron for each of the remaining three. 3. All of them (Don't for get the elctron ... Here is the full molecular orbital diagram for N 2. Now we add the 10 electrons, 5 from each nitrogen atom. Note that the bottom sigma symmetry orbital is strongly bonding, the top one is strongly antibonding, and the 2 in the middle are only weakly bonding and antibonding, respectively. The orientation in space of an atomic orbital is associated with... the spin quantum number (ms). ... The orbital diagram for a ground-state nitrogen atom is 1s 2s 2p A ↿⇂ ↿⇂ ↿ . ↿ . ↿ ... C ↿⇂ ↿⇂ ↿ . ↿ . ↿ D ↿⇂ ↿⇂ ↿⇂ ↿ . ↿ E ↿⇂ ↿⇂ ↿⇂ ↿⇂ ↿ A. The orbital diagram for a ground state ...
Atomic Orbital Diagrams: These are also known as electron-in-a-box diagrams. This is a simplified diagram of how the electrons are arranged within the orbitals for a particular atomic species. In a chlorine atom which subshells contain. Draw the atomic orbital diagram for chlorine. An orbital diagram is a sketch which shows electron population in atomic orbitals with the electrons. Create the atomic orbital diagram for chlorine. Create the atomic orbital diagram for nitrogen. The p orbital can hold up to six electrons. Answer to: create the atomic orbital diagram for nitrogen. By signing up, you'll get thousands of step-by-step solutions to your homework... MO Diagram for HF The AO energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The F 2s is nonbonding. H-F nb σ σ* Energy H -13.6 eV 1s F -18.6 eV -40.2 eV 2s 2p So H-F has one σ bond and three lone electron pairs on fluorine
Electron Configuration For Nitrogen Ion. The atomic number of nitrogen is 7, the element nitrogen was discovered by a Scottish physician, Danial Rutherford. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element Nitrogen.
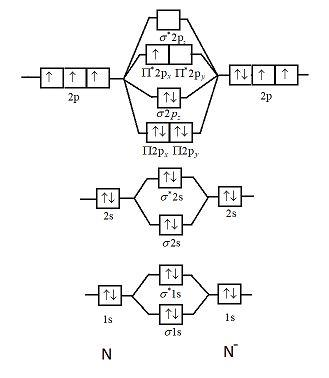
The molecular orbital energy level diagram of He 2 (hypothetical) is given in Fig. Here, N b = 2 and N a = 2. Bond order = N b -N a / 2 = 2-2 / 2 = 0. As the bond order for He 2 comes out to be zero, this molecule does not exist. 3. Nitrogen molecule (N 2). The electronic configuration of nitrogen (Z=7) in the ground state is 1s 2 2s 2 2p 1x 2p ...
The atomic number of nitrogen is 7 and its symbol is 'N'. The standard atomic mass of nitrogen is 14.006. The period of nitrogen is 2 and nitrogen is a p-block element. The electron configuration of nitrogen(N) and the orbital diagram is the main topic of this article.
Oxygen electron configuration is 1s 2 2s 2 2p 4.The period of oxygen is 2 and it is a p-block element. This article gives an idea about the electron configuration of oxygen(O) and orbital diagram, period and groups, valency and valence electrons of oxygen, bond formation, compound formation, application of different principles. The eighth element in the periodic table is oxygen.
Atomic no. Orbital Diagram of All Elements Diagrams; 1: Orbital diagram of Hydrogen (H) 2: Orbital diagram of Helium (He) 3: Orbital diagram of Lithium (Li) 4: Orbital diagram of Beryllium (Be) 5: Orbital diagram of Boron (B) 6: Orbital diagram of Carbon (C) 7: Orbital diagram of Nitrogen (N) 8: Orbital diagram of Oxygen (O) 9: Orbital diagram ...
Question: a) Complete the molecular orbital diagram for NF, nitrogen monofluoride below. Electrons can be added to the orbitals by clicking the orbital. Additional clicks will change the occupation of the orbital. Be sure to fill in both the atomic and molecular orbitals. Note: atomic orbitals are not shown. 629 2p 2p Tap 028 02 2s 2s 025 N F b ...
The figure below illustrating orbital diagrams for nitrogen is similar to the orbital. 年4月2日 You want electron configuration. Atomic # is the number of electrons that a particular element has. Here's the order of the energy shells. Orbital filling diagrams essentially just turn this big list of electron locations .
To see this video, other videos, chemistry education text, and practice problems visit my website. Website is 100% FREE to use.http://scientifictutor.org/
Answer to Create the atomic orbital diagram for nitrogen.
Figure 7.5.4. This orbital energy-level diagram shows the sp hybridized orbitals on Be in the linear BeCl 2 molecule. Each of the two sp hybrid orbitals holds one electron and is thus half filled and available for bonding via overlap with a Cl 3p orbital. When atomic orbitals hybridize, the valence electrons occupy the newly created orbitals.
About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators ...
Create the atomic orbital diagram for nitrogen. Start by adding the appropriate subshells. For example, boron is in the 2p block of the periodic table, and so you need to show the 2p subshell and everything below it. Next, click the orbitals to add electrons (represented as arrows). For boron, you would need to show a total of five electrons.
To create four equivalent bonding orbitals in carbon, the atomic wave functions, Y, for the 2s and three 2p orbitals are mixed mathematically, to give new molecular wave functions, and molecular orbitals. Mixing one s orbital with three p orbitals will produce four hybrid orbitals, called sp 3 orbitals.
6) A student attempted to create an orbital diagram for nitrogen, but they made a mistake. Briefly explain what they did wrong ( Subshell electron capacity 10 TIL 22
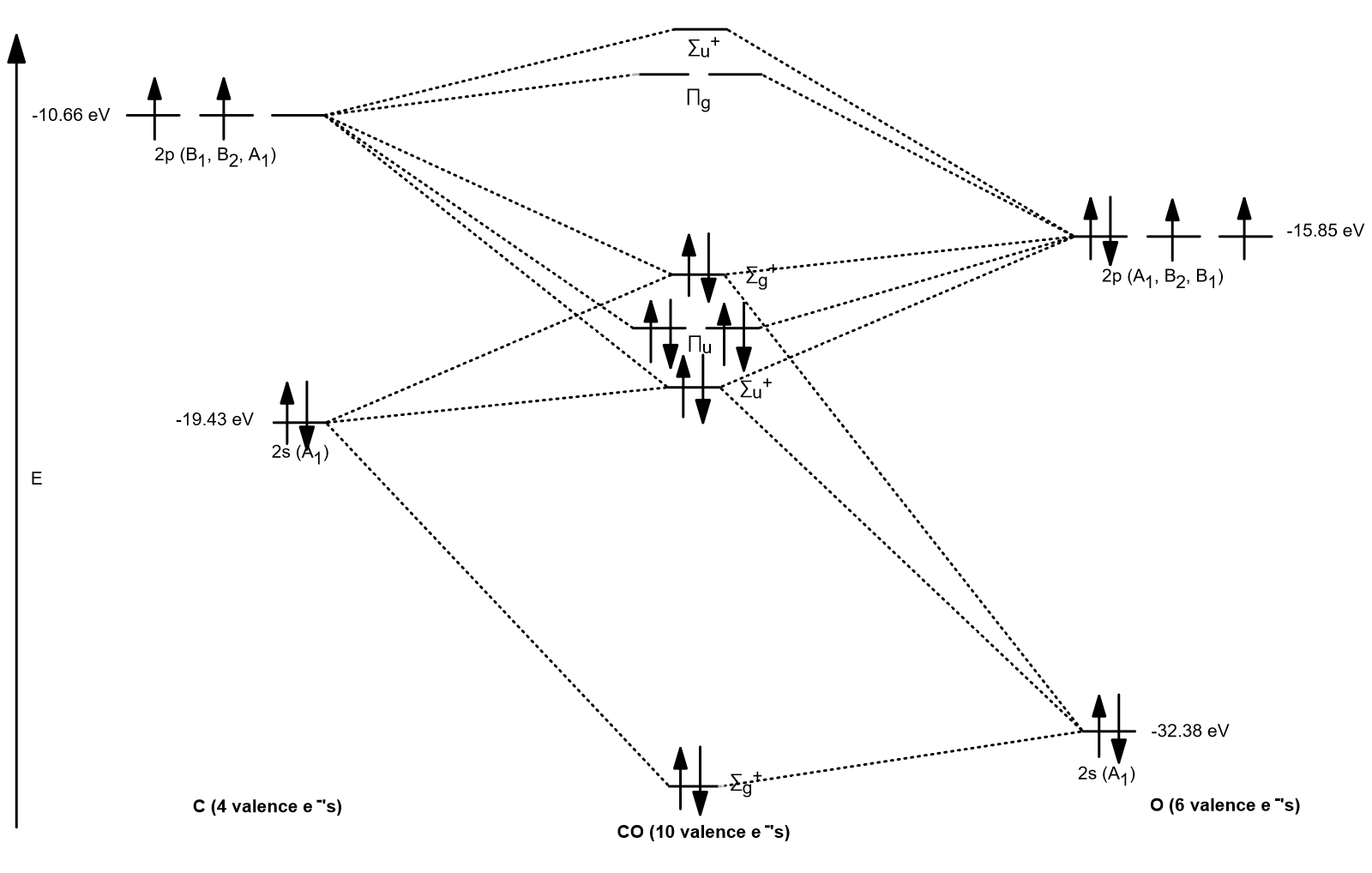
The correlation diagrams for nitrogen and carbon monoxide and the first are nearly parallel to the corresponding orbital energy curves. Bond order for N2 is 3; bond order for N2- is and bond order for N2+ is I have not included pictures of the MO diagrams that show the orbital energies. N2+ has less bond energy.
Create the atomic orbital Diagram for Nitrogen. solved create the atomic orbital diagram for nitrogen st answer to create the atomic orbital diagram for nitrogen start by adding the appropriate subshells for example boron is in the create the atomic orbital diagram for nitrogen in some cases we may need to slightly alter the design color or even accessories we want a new thought for it then ...
What is the atomic orbital diagram for nitrogen? The remaining three electrons will go in the 2p orbital. Therefore the N electron configuration will be 1s22s22p3. The configuration notation for Nitrogen (N) provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of the Nitrogen atom.
The choice A accurately specifies and illustrates the orbital diagram of a Nitrogen atom with 7 electrons. Based on the number of electrons in a Nitrogen atom, there are two energy levels, the s and p sub-levels: Nitrogen = 2, 5 . The first energy level, S will take up two electrons with opposite spin.



















0 Response to "37 create the atomic orbital diagram for nitrogen."
Post a Comment