40 incongruent melting phase diagram
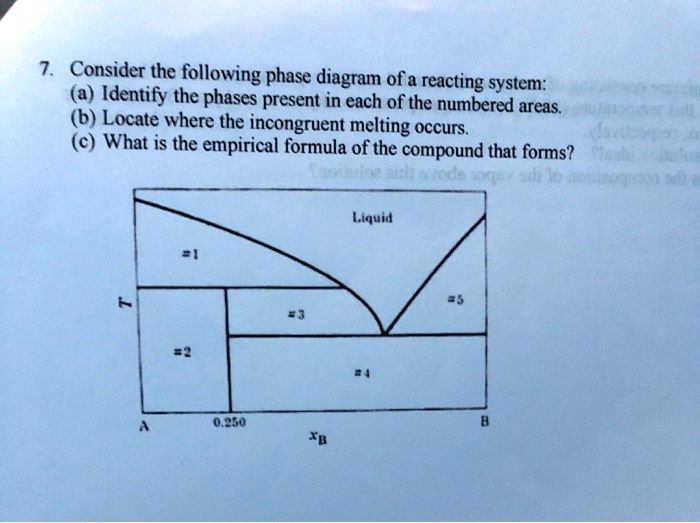
Incongruent melting - WikiMili, The Best Wikipedia Reader Congruent melting; Incongruent transition; Phase diagram; Related Research Articles. Magma is the molten or semi-molten natural material from which all igneous rocks are formed. Magma is found beneath the surface of the Earth, and evidence of magmatism has also been discovered on other terrestrial planets and some natural satellites. Besides ... D4a Indicate on the phase diagram in Fig 54 Fig 53 10th ... 5D.4(a) Indicate on the phase diagram in Fig. 5.4 (Fig. 5.3, 10th; Fig. 5.66, 9th) the feature that denotes incongruent melting. What is the composition of the eutectic mixture and at what temperature does it melt? Fig. 5.4: 5D.4(b) Indicate on the phase diagram in Fig. 5.5 (Fig. 5.4, 10th; Fig. 5.67, 9th) the feature that denotes
1.: Example of a binary phase diagram with an incongruent ... Download scientific diagram | 1.: Example of a binary phase diagram with an incongruent melting phase γ. Inset: The distribution coefficient is set constant by assuming straight liquidus and ...

Incongruent melting phase diagram
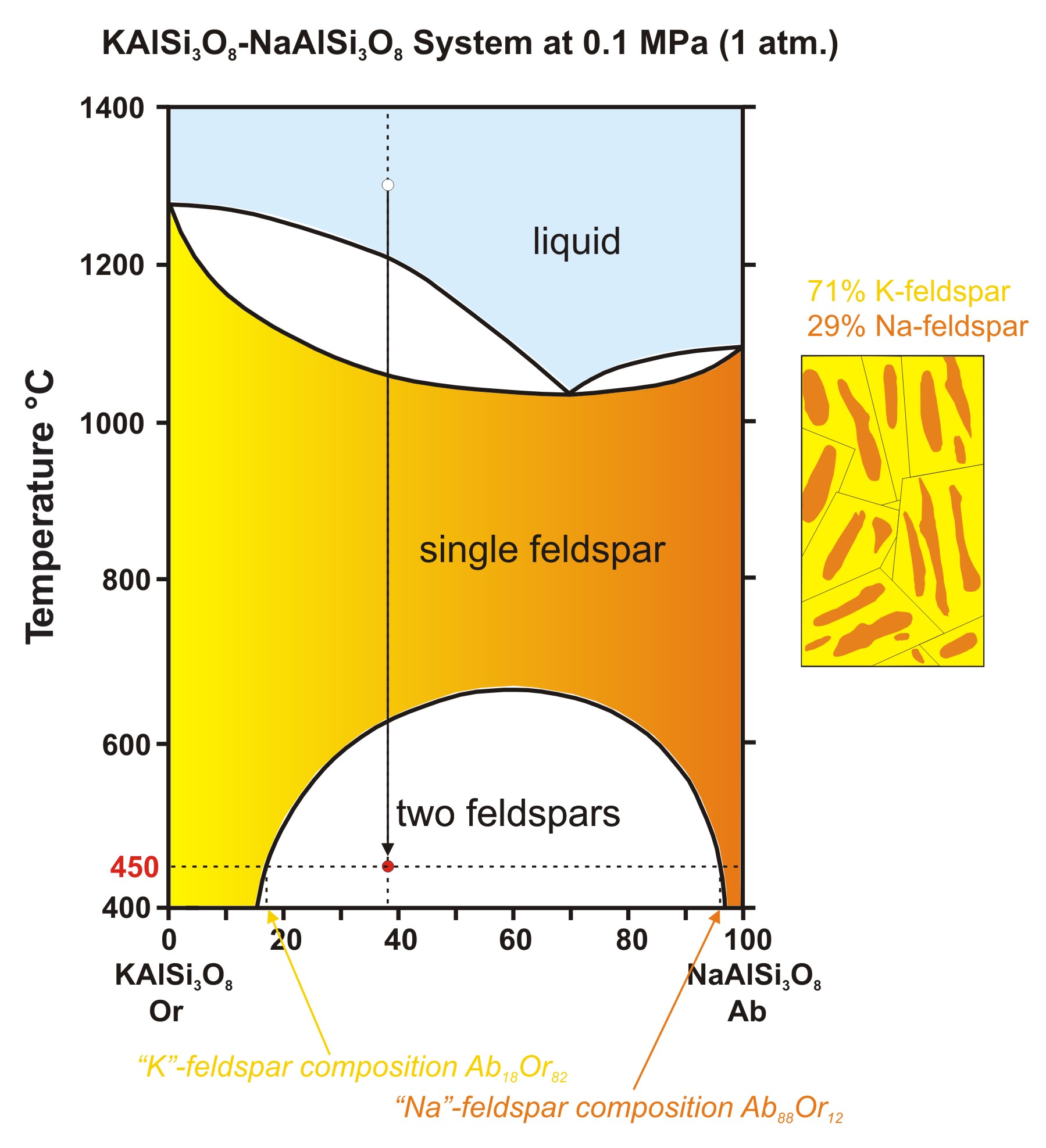
Formation of Compounds with Congruent & Incongruent ... The phase diagram has four maxima corresponding to the formation of four hydrates. Point C (37°C). E (32.5°C) and I (78.5°C) represent the congruent melting point of dodeca, hepta, penta and tetra hydrates respectively. The congruent melting point of a salt hydrate is also known as the dyestectic point. Copper sulphate water vapour system Identifying congruent vs incongruent melting intermediate ... Phase diagrams commonly contain intermediate compounds. There are both incongruent and congruent melting compounds. An incongruent melting point will end in ... Phase Equilibria and Phase Rule (Part - 2 ... 1 Na-K system This is a two-component system having incongruent melting point. The melting points of sodium and potassium are 97.8°C and 63.8°C respectively which are shown in the phase diagram in Fig. 2.4. Both elements chemically combine together in the ratio of 2:1 to form a compound Na 2 K.
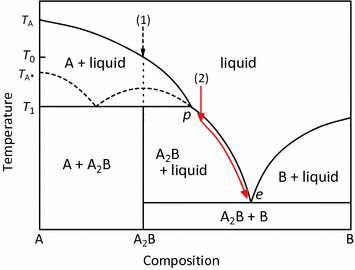
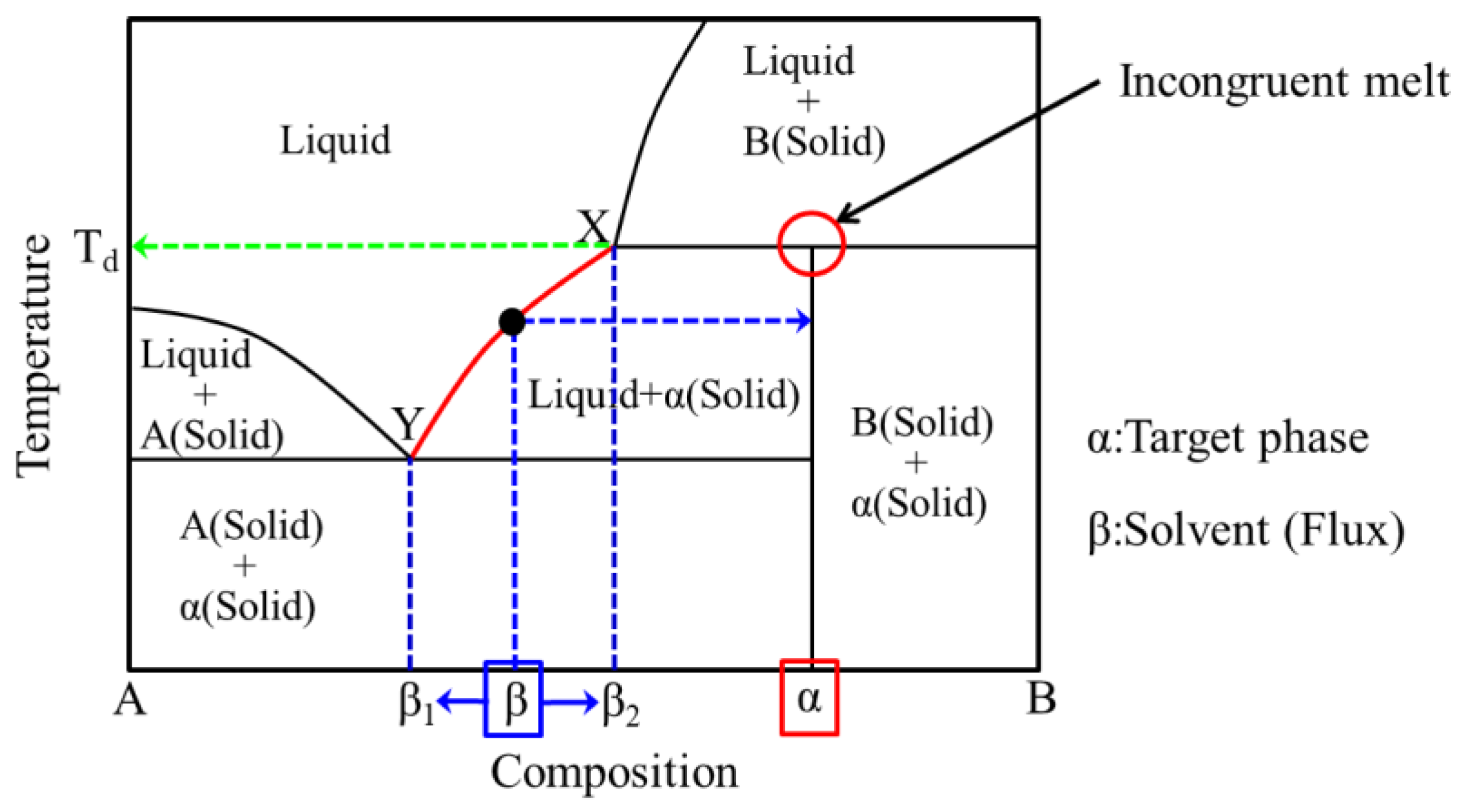
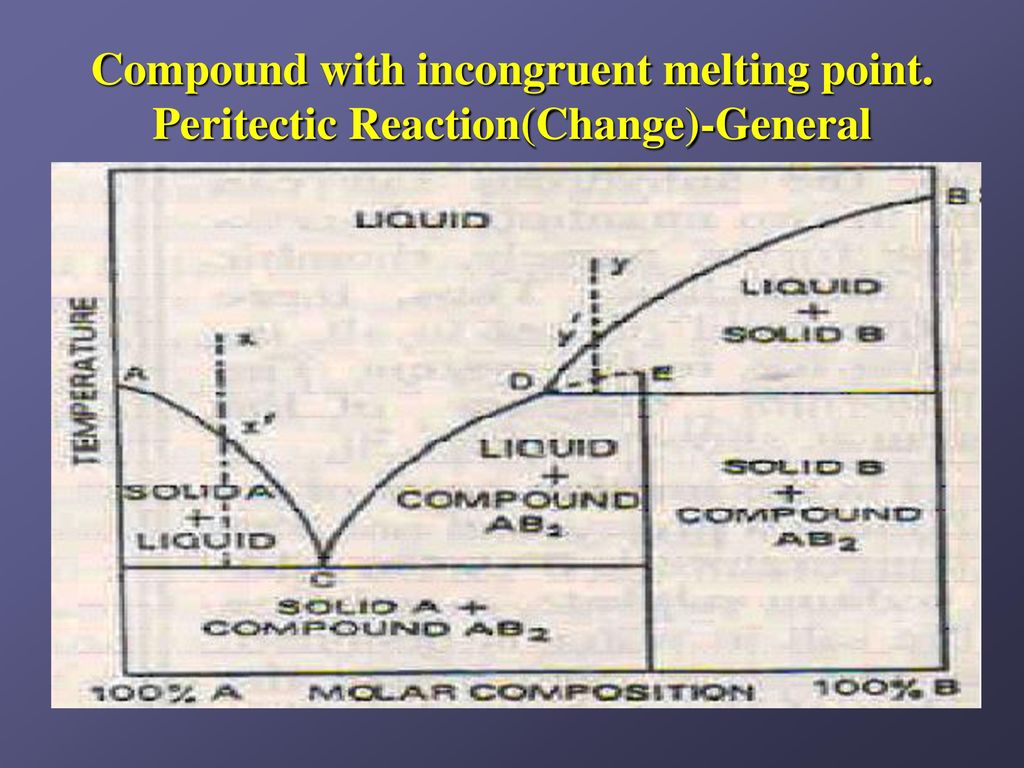
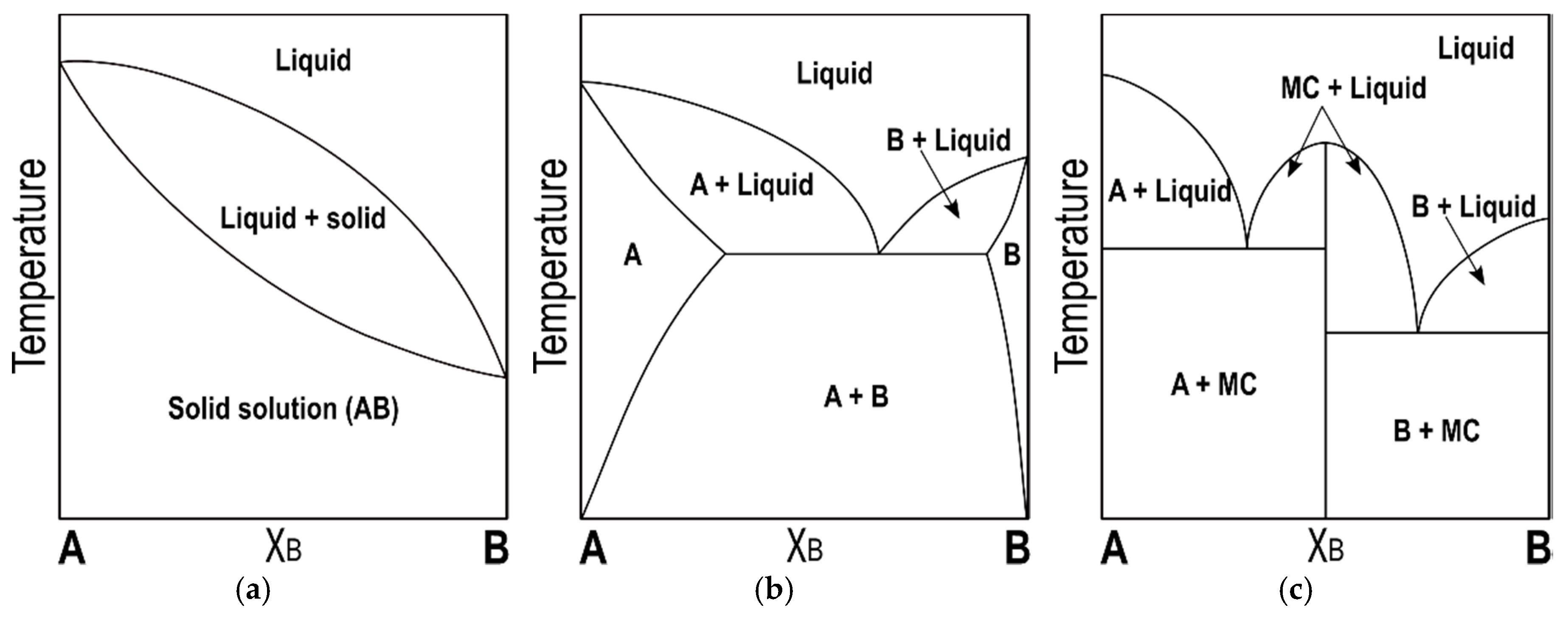
Incongruent melting phase diagram. Congruent Melting Point || Phase Rule || Phase Diagram ... For Complete Courses Download The App Chemistry Untold :- Solid State Complete Course :- http... Congruent melting - WikiMili, The Best Wikipedia Reader Congruent melting occurs during melting of a compound when the composition of the liquid that forms is the same as the composition of the solid. It can be contrasted with incongruent melting.This generally happens in two-component systems.To take a general case, let A and B be the two components and AB a stable solid compound formed by their chemical combination. Congruent and Incongruent Melting In Binary and Ternary The intermediate compound in this diagram (XY 2) however is incongruently melting. Incongruent melting is the temperature at which one solid phase transforms to another solid phase and a liquid phase both of different chemical compositions than the original composition. This can be seen in this diagram as XY 2 melts to Y and liquid. more the peritectic reaction is an example of incongruent ... more, the peritectic reaction is an example of incongruent melting for an inter-mediate phase. Concept Check 9.6 The figure on the next page is the hafnium-vanadium phase diagram, for which only single-phase regions are labeled.
Phase Equilibria and Phase Rule (Part - 2 ... 1 Na-K system This is a two-component system having incongruent melting point. The melting points of sodium and potassium are 97.8°C and 63.8°C respectively which are shown in the phase diagram in Fig. 2.4. Both elements chemically combine together in the ratio of 2:1 to form a compound Na 2 K. Identifying congruent vs incongruent melting intermediate ... Phase diagrams commonly contain intermediate compounds. There are both incongruent and congruent melting compounds. An incongruent melting point will end in ... Formation of Compounds with Congruent & Incongruent ... The phase diagram has four maxima corresponding to the formation of four hydrates. Point C (37°C). E (32.5°C) and I (78.5°C) represent the congruent melting point of dodeca, hepta, penta and tetra hydrates respectively. The congruent melting point of a salt hydrate is also known as the dyestectic point. Copper sulphate water vapour system











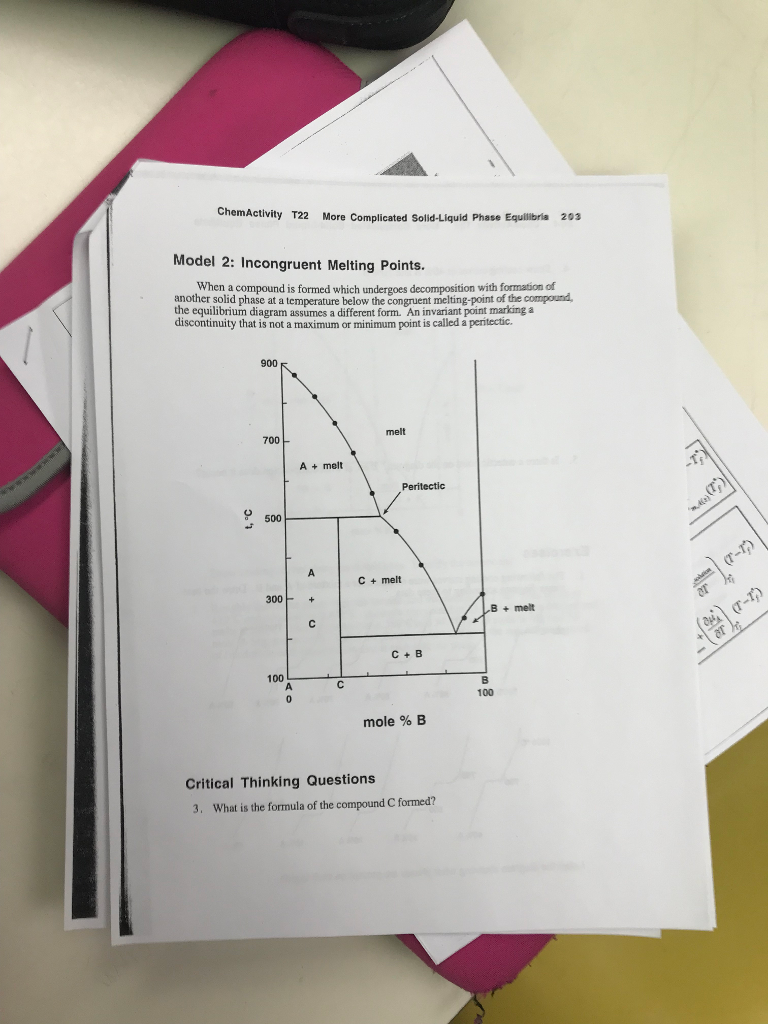



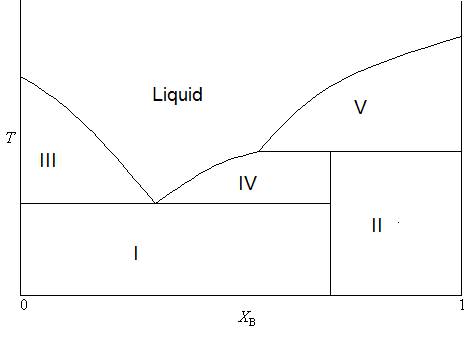
0 Response to "40 incongruent melting phase diagram"
Post a Comment